by W.A. Steer PhD
![]()
by W.A. Steer PhD
| Back to contents | About... |
Ā
![]()
In graphics, printing, and lighting we often talk about a 'white-point' in terms of a temperature, e.g. 5000K or 6500K. This page explains the origin of this description, as well as showing some colourimetrically-accurate colour-temperature plots. I round off with a few words about illuminants and colour-constancy.
It is well known that when an object, such as a lump of metal, is heated, it glows; first a dull red, then as it becomes hotter, a brighter red, then bright orange, then a brilliant white. Although the brightness varies from one material to another, the colour (strictly spectral distribution) of the glow is essentially universal for all materials, and depends only on the temperature. In the idealised case, this is known as 'black body' or 'cavity' radiation, and is described by Planck's Radiation Law:
Spectral energy density, U(λ,T) = 8πhcλ-5 / ( ehc/λkT-1 )
where
λ is the wavelength, in metres
T is the temperature in Kelvin (add 273 degrees to Celcius temperatures to get Kelvin)
h = 6.626×10-34 J·s [Planck's constant]
k = 1.381×10-23 J·K-1 [Boltzmann's constant]
c = 3.0×108 m·s-1 [speed of light]
For any more detail, see an undergraduate-level Physics textbook!
The figure below illustrates the relative amounts of energy at each wavelength across the visible spectrum, for a 'black body' at 3200K, 5000K, 6500K, and 9300K. A conventional incandescent light bulb emits light (and heat!) from a tungsten filament heated to high temperature by passing an electric current through it. Spectral distribution (including colour) of the light from a quartz-halogen lightbulb is similar to a black body source at 3200K, and is therefore well-approximated by the Planck Law. Light from the sun, measured in space, is close to black body radiation characteristic of temperatures around 5000-6500K.
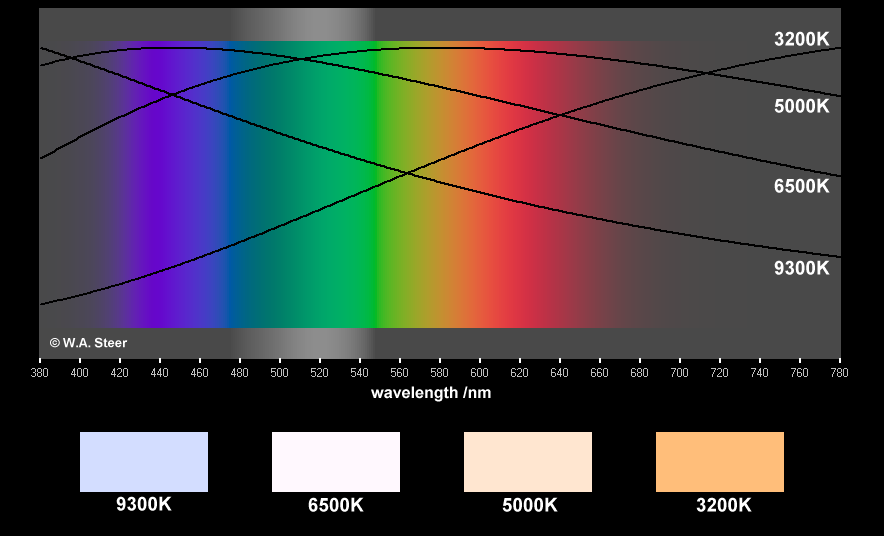
Black-body radiation curves (vertical scale is linear). If your computer and monitor adhere to the sRGB specification (your monitor colour-point should be
set to 'sRGB' or '6500K') then the sample colours should be colourimetrically-accurate.
Due to absorbtion and scattering in the Earth's atmosphere, the solar spectrum at the earth's surface is modified somewhat depending on the time of day and weather conditions. On a clear day direct light from the sun will contribute the primary, highly directional, illumination, while the whole dome of the sky will contribute a diffuse bluey ambient illumination. During a cold, frosty morning the scattered bluer components may be more dominant in defining the ambient illumination; perhaps counter-intuitively, this bluer light has a colour approximating to a black-body at a higher temperature (eg 10000K).
Our eyes readily adapt not only to different levels of illumination (daylight vs moonlight), but also to different tints in the colour of light. A non-luminous object (such as a piece of paper) is objectively agreed to be 'white' if it reflects all colours (visible wavelengths) approximately equally... which means it takes on the colour appearance of the ambient illumination. But unless we consciously think about it (or are photographers or colour-scientists), most people are happy to regard illumination from regular filament lightbulbs, sunlight (under varying conditions) and even fluorescent light as being 'white'! Colours which are generally 'acceptable' as 'white' are usually quite close to black body colours upwards of around 2500K. Consequently 'whites' for the purposes of graphics and photography are often specified in terms of a colour-temperature.
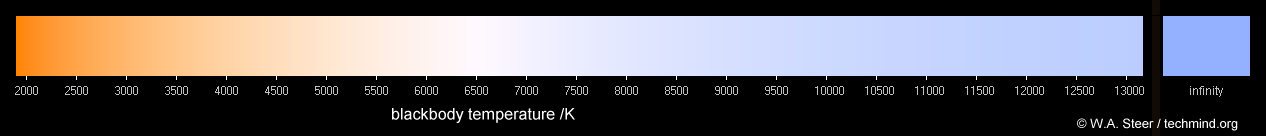
Colours of black body sources. Note that it is not physically possible to accurately mimic blackbody colours cooler than
1900K on a standard sRGB (or Rec.709) monitor; the red primary of the display is not a deep enough red!
Again these sample colours have been calculated for colourimetric accuracy. If your computer and monitor
adhere to the sRGB specification (your monitor colour-point should be set to 'sRGB' or '6500K', and your
brightness and contrast controls properly adjusted) the colours should be reasonably close.
As the temperature is increased beyond 10000K the colour gradually converges towards a pale sky-blue,
shown at the far right of the graphic as "infinity". I've included this colour for completeness;
it's of little practical significance since anything of such high temperature would be so blindingly
bright as to make its colour fairly irrelevant, also you'd be scorched by the ultraviolet radiation and
X-rays. Furthermore, at such temperatures I guess atoms would vaporize; you'd have a plasma and various
atomic emission (and absorption) lines in the spectrum too - so it wouldn't be a true black-body!




Black body sources (approximately any filament bulb or sunlight - but not fluorescent lamps, in general) emit a smooth distribution of wavelengths across the visible spectrum, which means that our eyes and visual system can reliably distinguish colours of non-luminous objects. Subconsciously we adapt to differing bias in the illuminant colour, and manage to perceive consistent colours in the artifacts we handle every day (food, clothes, etc) - despite wide variations in their absolute colour.
Artificial sources of light, in particular discharge lamps (sodium, mercury, xenon) and fluoresent lamps can have extremely spikey spectral distributions, and this means that their colour rendering properties are very poor (even if the overall perceived illuminant colour is close to a blackbody colour).
In professional lighting, a Colour Rendering Index, CRI (sometimes written Ra) is often quoted to indicate how accurately that light will portray colours relative to a blackbody source at the same nominal colour temperature. By definition, all backbody sources have a CRI of 100. Fluorescent lamps typically have CRIs in the range 55-85, with 80-85 being classed by the manufacturers as 'good' or 'very good' colour-rendering. I beg to differ! To my critical eyes, light from 'triphosphor' fluorescents with a CRI of 80-85 is ghastly; extremely flat and lifeless. By comparison, a nice incandescent quartz-halogen spotlight really gives vitality to whatever it shines on!
©2005-2008 William Andrew Steer